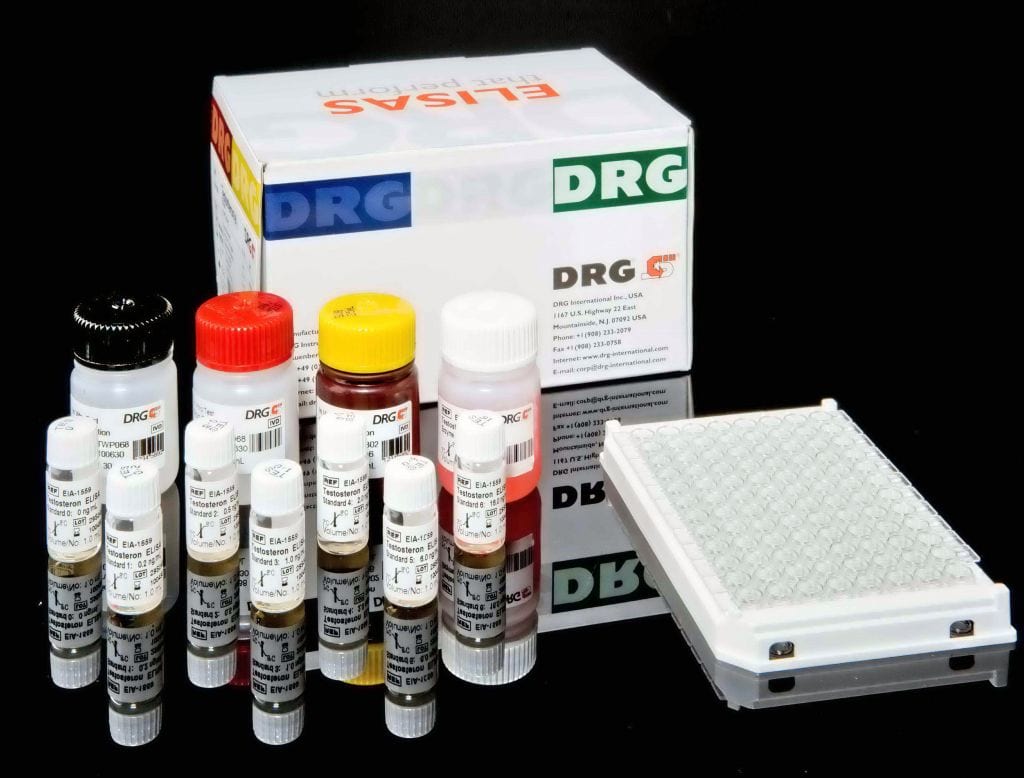
Description
The Chlamydia pneumoniae IgG-ELISA is intended for the qualitative determination of IgG class antibodies against Chlamydia pneumoniae in human serum or plasma (citrate).
The qualitative immunoenzymatic determination of IgG-class antibodies against Chlamydia pneumoniae is based on the ELISA (Enzyme-linked Immunosorbent Assay) technique.
Microtiter strip wells are precoated with Chlamydia pneumoniae antigens to bind corresponding antibodies of the specimen. After washing the wells to remove all unbound sample material horseradish peroxidase (HRP) labelled anti-human IgG conjugate is added. This conjugate binds to the captured Chlamydia pneumoniae-specific antibodies. The immune complex formed by the bound conjugate is visualized by adding Tetramethylbenzidine (TMB) substrate which gives a blue reaction product. The intensity of this product is proportional to the amount of Chlamydia pneumoniae-specific IgG antibodies in the specimen. Sulphuric acid is added to stop the reaction. This produces a yellow endpoint colour. Absorbance at 450 nm is read using an ELISA microwell plate reader.